Federal Judge Strikes Down Mandatory Cigarette Health Warning Labels
|
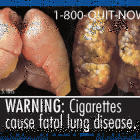
This week a federal judge ruled in favor of tobacco companies challenging a FDA requirement that would force cigarette manufacturers to place graphic warning labels on their products. “The government has failed to carry both its burden of demonstrating a compelling interest and its burden of demonstrating that the rule is narrowly tailored to achieve a constitutionally permissible form of compelled commercial speech," U.S. District Judge Richard J. Leon said. The ruling stems from a 2009 law passed by Congress that required the FDA to enforce new warning requirements, which included manufacturers placing color labels on their products which covered at least half of the packaging space, as well as on 20 percent of print advertisements for cigarettes. Last summer, the FDA unveiled nine warning labels – among them, photographs of charred lungs and corpses – that were expected to be placed on all cigarette packages in the United States by September 2012. Last year, Judge Leon allowed a preliminary injunction which prevented the mandatory warnings from being placed on tobacco products, a decision challenged by the Obama administration and currently awaiting a U.S. Court of Appeals for the District of Columbia Circuit ruling.

